|
10/7/2023 0 Comments Standard molar entropy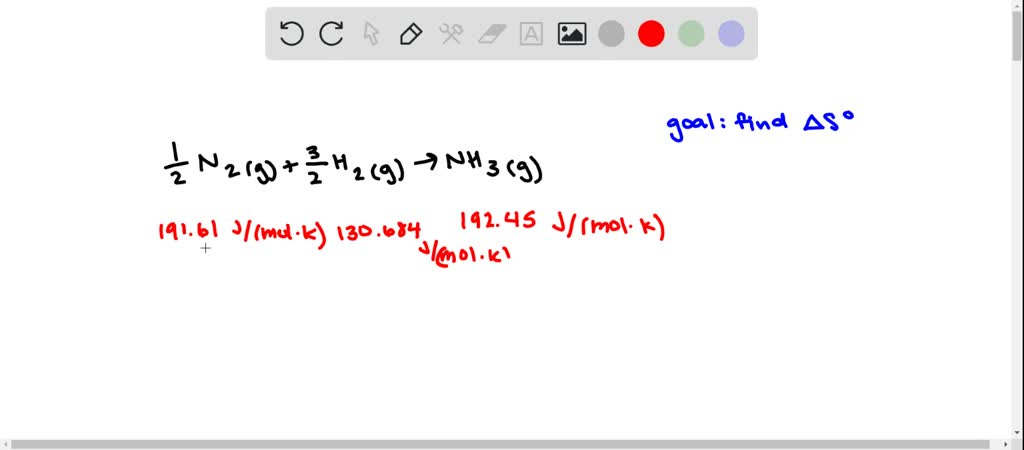
Where P is equilibrium vapor pressure in k Pa, and T is temperature in kelvins.įor T = 273 K to 333 K: A = 7.2326 B = 1750.286 C = 38.1.įor T = 333 K to 423 K: A = 7.0917 B = 1668.21 C = 45.1.ĭata in the table above is given for water–steam equilibria at various temperatures over the entire temperature range at which liquid water can exist. 
Vapor pressure formula for steam in equilibrium with liquid water: log 10 P = A − B T − C, The values below 0 ☌ refer to supercooled water.Įlectrical conductivity of highly purified water at saturation pressure Temperature, ☌ Liquid physical properties Temperature dependence of the surface tension of pure water Temperature dependence of the density of ice and water Velocity of sound in water Thermodynamic properties Phase behaviorħ5.97 J/(mol K) and 4.2176 J/(g Structure and properties Structure and propertiesĤ92.215 kJ/mol O–H bond dissociation energy Excited state at 3.0 eV,81WU /TIE.This page provides supplementary data to the article properties of water.įurther comprehensive authoritative data can be found at the NIST Webbook page on thermophysical properties of fluids. Gas phase Relative to HBr, reevaluated with current HBr acidity. Gas phase Entropy change calculated or estimated Mīy formula: NO 3 - + H + = HNO 3 Quantity Reaction search pages in place of the enumerated reaction Go To: Top, Gas phase thermochemistry data, Phase change data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notes Chickos Enthalpy of vaporization Δ vapH (kJ/mol) Go To: Top, Gas phase thermochemistry data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesĭata compiled by: William E. 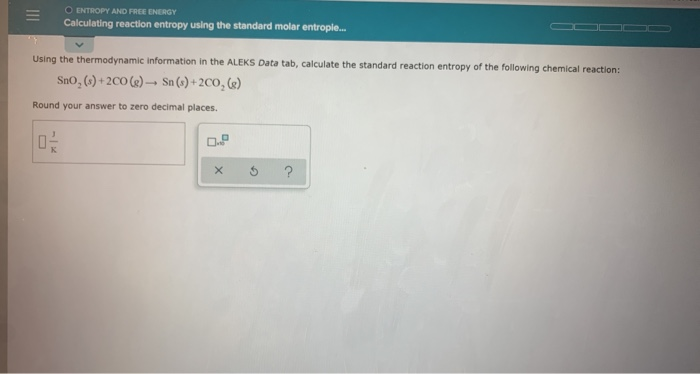
Requires a JavaScript / HTML 5 canvas capable browser. Gas Phase Heat Capacity (Shomate Equation) Secretary of Commerce on behalf of the U.S.A. Go To: Top, Phase change data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesīy the U.S. Your institution may already be a subscriber.įollow the links above to find out more about the data With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
